Please contact us at info@sofpromed.com if you need a CRO to conduct a soft tissue sarcoma clinical trial in the US
Are you a small or midsized biotech company planning a clinical trial in soft tissue sarcoma? If your answer is yes, then you may be interested in selecting a reliable full-service contract research organization (CRO) to help you manage all the different stages of the research life-cycle, from study initiation to study closure.
Since 2012, Sofpromed has been offering its managing services to many biotech companies across Europe and the United States. Our oncology-focused CRO has great experience managing personnel, resources, as well as all the activities involved in the conduct of clinical investigation. Therefore, on account of its experience and professionalism, Sofpromed is potentially the best partner for small-sized biotech companies.
Soft Tissue Sarcoma (STS): An Overview
Soft tissue sarcoma (STS) is the term used to refer to a malignant rare cancer type of mesenchymal origin. STS start in the soft or connective tissues that support or connect the body. They can develop in fat, nerves, muscles, bones, tendons and ligaments, blood vessels, lymph vessels, cartilage, the tissues around the joints, and other soft or conjunctive tissues.
This type of cancer is an uncommon aggressive group. The incidence of soft tissue sarcoma is considerably low as it only represents 5 cases per 100,000 people per year. Many soft tissue sarcomas are very rare and there are also many uncertain types.
These are the most common types of soft tissue sarcoma:
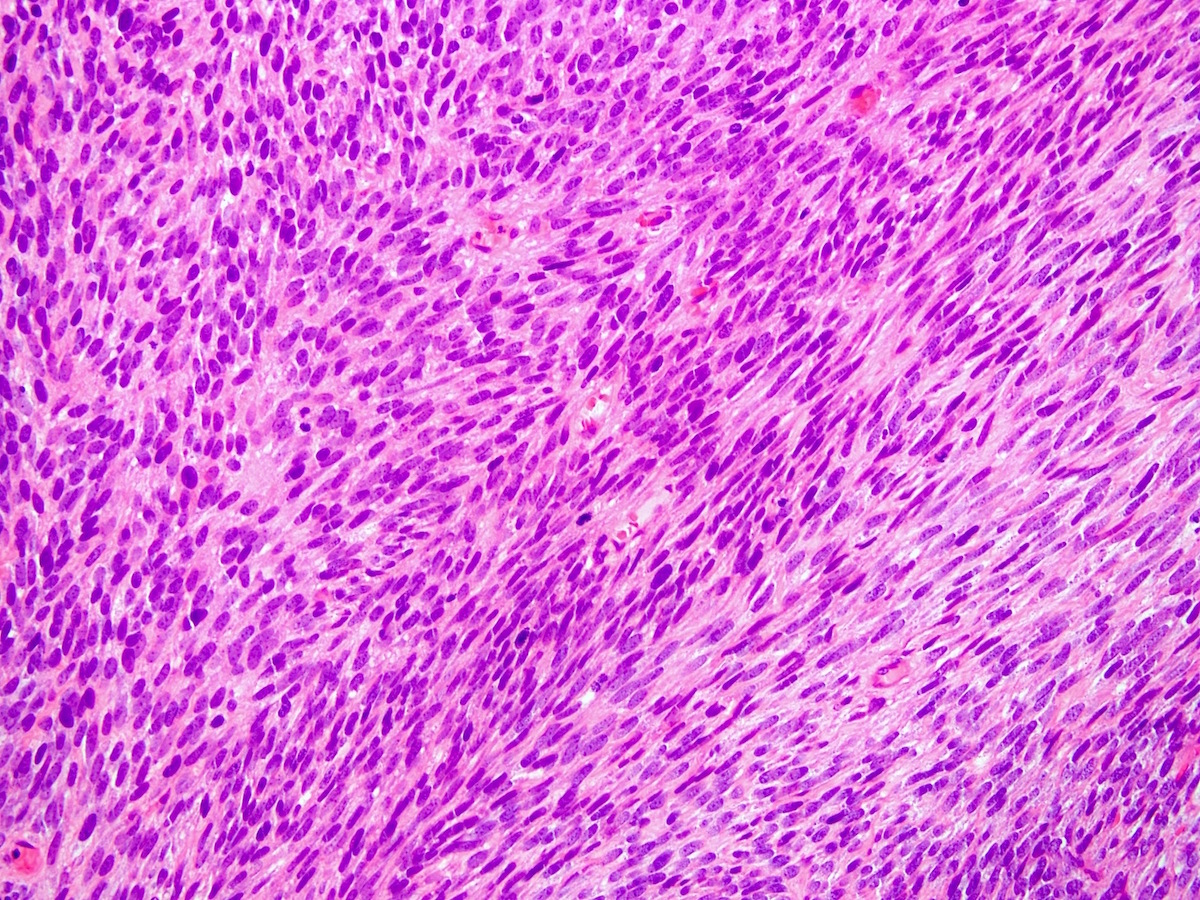
- Leiomyosarcomas: this is the most common type of soft tissue sarcoma among adults. It grows in the smooth muscles, which is the type of muscle that people use involuntarily. It can arise in the abdomen, digestive system, large blood vessels, womb (uterus), limbs and skin.
- Liposarcomas: this rare type of cancer develops from the fat cells, and forms about 20% of soft tissue sarcomas in adults. Although it can grow almost anywhere in the body, the most common places include the muscles of the abdomen or the limbs. It is most frequent in legs.
- Fibrosarcomas: this very rare tumor represents about 1% of soft tissue sarcomas, and it is generally diagnosed in adults between the ages of 20-60. They start in fibrous connective tissues, such as deep tissues of the legs or arms, abdomen and chest.
- Rhabdomyosarcomas: they begin in the striated muscles, the term used to refer to the muscles that can be controlled (voluntary muscles). Some examples of this kind of tumor are embryonal, alveolar and pleomorphic rhabdomyosarcomas.
The symptoms of soft tissue sarcoma are not always perceivable in its early stages. In fact, most patients with this type of cancer present with symptoms once the tumor has grown, usually in an arm or leg, or when it has spread (metastasized) to other parts of the body and presses on nearby organs, muscles, nerves or blood vessels.
Soft tissue sarcoma can manifest itself in widely different ways, mainly depending on where the tumor develops, which makes its detection a real challenge. However, there are certain signs of the disease that should be paid attention to. Here is a list of the most common symptoms of soft tissue sarcoma:
- A new lump or a lump that is getting bigger
- Acute pain in the abdomen
- Swelling in the belly or under the skin
- Blood in your vomit or stool
- Feeling full and constipation
- Difficulty breathing
The identification of these signs is essential for early detection of soft tissue sarcoma. If any of these symptoms appears, it is very important to make an appointment with the doctor to carry out an accurate diagnosis and thus ensure the earliest possible care and treatment.
High Risk Groups for Soft Tissue Sarcoma in the United States
Soft tissue sarcoma is a rare type of cancer as it only represents 0.7% of all new cancer cases in the world. Although it can affect people of all ages, ethnicities and genders, men are more prone to develop STS than women, and the risk of developing it increases as people grow older.
According to the American Cancer Society, an estimated 13,190 people in the United States will be diagnosed with STS in 2022, of whom 5,130 will eventually die from this illness. [1] Hence, in comparison with other types of cancer —such as lung cancer, which is expected to be the cause of death of 608,570 people in the US this year—, soft tissue sarcoma does not represent a huge impact on mortality in the country.
It has been proven that most soft tissue sarcomas develop in individuals with no apparent risk conditions. However, there are a series of factors associated with a higher risk of STS. People may be at risk of developing soft tissue sarcoma if they have:
- Been exposed to certain chemicals
- Received previous radiotherapy treatment
- Some types of rare genetic conditions
- A weakened immune system
- Chronic lymphedema
Moreover, there are also considerable racial disparities, which are shown by the fact that African American individuals are more likely to present with larger size tumors and, as a result, the life expectancy of black patients is shorter than it is for their white counterparts. For the sake of comparison, while the 5-year survival rate for black patients with STS is 61.4%, the percentages for white and Asian patients are 66.9% and 69.9%, respectively. [2]
In addition, black patients are less likely to undergo surgical resection, receive chemotherapy or radiotherapy, or be insured than their white peers, which makes them more vulnerable. Nevertheless, race by itself is not an independent predictor of a poor prospect of survival. If soft tissue sarcoma in black patients has a worse prognosis, it is due to the socioeconomic status of these minority populations.
Actually, high rates of poverty, as well as below-average education and income levels, put them at a comparative disadvantage. For this reason, in order to break social barriers, it is crucial to provide this group of at-risk patients with timely care and appropriate treatment.
Current Treatments and Medical Need for Patients with Soft Tissue Sarcoma
Due to the low incidence rate of soft tissue sarcoma, unfortunately not much investigation has been conducted to find new and more effective treatments for patients with this disease. In this section, we will take a close look at the traditional and newer therapies currently used to alleviate the symptoms and reduce the morbidity caused by this cancer.
Once a person experiences any of the symptoms associated with soft tissue sarcoma, it is important to make an appointment with your primary care physician. This professional will examine the patient and will then determine whether or not to refer him/her to a sarcoma medical oncologist for consultation.
The oncology expert will review the patient’s full medical history and make an accurate diagnosis of the disease. If the sick individual is eventually diagnosed with soft tissue sarcoma, the specialist will take the time to explain to the patient everything he/she needs to know about his/her condition, answer any questions that may arise, and help the patient decide the most appropriate therapeutic plan for his/her specific situation.
Moreover, it may be advantageous to request a second opinion, that is, advice from a second expert. A second opinion may be useful to get more information, hear a different point of view, and ensure that both the first diagnosis and the treatment option chosen are correct.
The following is a brief presentation of the treatments used for soft tissue sarcoma.
Surgery
Surgery continues to be the main treatment for soft tissue sarcoma, and more particularly for leiomyosarcoma (LMS). In this medical intervention, which consists of removing the tumor in a clinical operation, the whole tumor, along with a small part of healthy tissue around the tumor, will be surgically removed in order to prevent the cancer from returning.
The type of surgery needed will vary depending on the location and size of the tumor. Limb-sparing surgery is the most conventional form of surgery to treat STS. It involves drawing tissue from the abdomen, limbs, head and neck, among other parts of the body. Only in a few cases, especially if the tumor has spread to nearby tissues, will amputation be required.
In some cases, a minor surgical operation is sufficient to remove the disease while, in other cases, it is necessary to perform a major surgical operation that may affect the proper functioning or physical appearance of the treatment area. For example, removing a large amount of tissue may have an effect on how well a patient can use his/her legs or arms.
Sometimes, surgery is used along with other therapies. For example, radiation and chemotherapy may be given after surgery (adjuvant theory) with the aim to help eliminate any remaining cancer cells. Alternatively, the patient can also receive neoadjuvant treatment, which consists of using chemo or radiation to shrink the tumor prior to surgical removal.
Radiotherapy
Radiation therapy is a key part of STS treatment. It uses high-energy rays to kill cancer cells with as little harm as possible to healthy cells. This type of treatment works to shrink or destroy the tumor and minimize the chance of cancer spread or recurrence.
Radiotherapy is the primary treatment for rhabdomyosarcoma (RMS), and it is often used in combination with chemotherapy. Radiation therapy is frequently used when some cancer cells are still left after surgery, or as an alternative to surgery in those cases where it would result in any loss or anomaly of an important organ. [3]
Patients receiving radiotherapy may experience side effects, which usually disappear little after the treatment finishes. Possible complications include hair loss, skin redness, nausea and fatigue. Before patients begin therapy with radiation, the clinical oncologist will have previously informed them of the risks involved so that they become aware of them in advance.
Chemotherapy
Chemotherapy uses cytotoxic drugs to treat various cancers, among which is soft tissue sarcoma. These anticancer drugs are usually administered as an injection into a vein (intravenously), but they can also be taken in tablet or capsule form.
There are different chemo drugs to treat soft tissue sarcoma. The most commonly used drugs are doxorubicin and ifosfamide, although other drugs —epirubicin, gemcitabine, docetaxel, trabectedin— may also be given, either on their own or in combination with others. Some of these drugs may be given to treat STS before surgery (neoadjuvant treatment), right after it (adjuvant treatment), or after tumor relapse (advanced or metastatic setting).
Because of its efficacy, chemotherapy is one of the first choices for metastatic disease. This type of therapy also plays a significant role in patients with a worse prognosis of non-metastatic cancer.
Cancer patients undergoing chemotherapy for soft tissue sarcoma can also experience side effects that may impair their health. Some examples include hair loss, increased risk of infection, mouth or throat sores, changes to your taste, anemia, nausea and fatigue.
Targeted therapy
Targeted therapies are a more recent form of drug therapy. These drugs attack parts of cancer cells that differ from the ones composing healthy cells. All the different targeted therapies interfere with the way cancer cells grow, replicate, work or interact with other cells of the body.
These therapies are usually given as part of clinical trials for investigators to study the specific genetic differences of tumors and discover new molecular targets for anticancer drug development. Nevertheless, as knowledge is acquired on the pharmacological properties of these drugs, targeted therapies are becoming increasingly important to treat soft tissue sarcoma.
Examples of current targeted drugs for soft tissue sarcoma are larotrectinib, sorafenib, sunitinib, regorafenib, and tazemetostat. Regorafenib and sunitinib are the most powerful tyrosine kinase inhibitors (TKIs) approved for gastrointestinal stromal tumors (GISTs), while pazopanib is the most important drug employed for non-GIST STS. [4]
Some common side effects of targeted therapies are muscle or joint pain, high blood pressure, nausea, diarrhea, loss of appetite, and tiredness. It may be difficult to cope with all these side effects, yet most of them are temporary and disappear when the treatment finishes.
In some cases, patients may also be able to participate in cutting-edge clinical trials in which targeted therapies are used. This is known as precision medicine, that is, a modern approach for disease treatment that considers biological factors such as genetics, environment and lifestyle. Precision medicine facilitates personalized care for specific groups of patients.
Current medical need for new drugs and therapies
Traditional treatments, such as chemo or radiation therapy, have proven to be effective to relieve the symptoms caused by STS and improve the patient’s quality of life to a certain degree. However, given the rare and diverse nature of this deadly cancerous disease, there is a major current need for new and multidisciplinary approaches to combat refractory or relapsed cancer.
As a result, more and more biotechnology and pharmaceutical companies are willing to discover new drugs and therapies for soft tissue sarcoma. To this end, scientists are looking at new combinations of medicines, doses, timing, and maintenance treatments aimed at helping patients with STS live longer and better lives. Certainly, there is a great scope for significant exploration to be made in this scientific field.
How Can Sofpromed Help Biotech Companies Conducting Clinical Trials in Soft Tissue Sarcoma?
If you are a biotech company planning a clinical trial in soft tissue sarcoma, Sofpromed can become a strategic partner. Our oncology-focused CRO stands out for its deep knowledge of certain cancers, including soft tissue sarcomas, expertise and devoted commitment to offer products and services of high technical quality and innovation.
Sofpromed’s staff comprises fully qualified and certified clinical research associates (CRAs), statistical programmers, data managers, and many other professionals specialized in oncology, with a strong background in sarcomas. All of them have, on average, ten years of experience giving support to biotech and pharma companies conducting clinical research studies.
We are a fast-growing company with long-standing experience managing clinical trials internationally. In fact, an increasing number of biotech companies located in Europe, as well as in the United States, decide to put their trust in our services and contact us for assistance.
Due to this active international engagement, we have close connections with prestigious hospitals and clinics recruiting sarcoma patients. Therefore, we can provide biotech companies with the best sites to run clinical trials in these rare tumors, thus saving their time and effort. This proves particularly useful if there is a need to recruit patients with ultra rare tumor subtypes.
Furthermore, thanks to our extensive network of contacts, we can also give access to key opinion leaders (KoL). These sarcoma specialists of great renown will offer their informed opinions, which can add great value to the design of the clinical protocol. Certainly, the perspectives of these medical experts will make clinical trials more successful.
We are also very knowledgeable of the laws and regulations governing drug development and manufacturing. As a result of this awareness and our great data management capabilities, we can generate robust data for regulatory authorities.
Sofpromed offers a comprehensive range of services, some of which are described briefly below:
- Clinical operations: our qualified and experienced clinical research associates (CRAs) are responsible for initiation visits, onsite monitoring visits, and clinical site management, among many other tasks.
- Biometrics: the main goal of our team of expert statistical programmers is to ensure that all data collected during a clinical study is used to its maximum potential.
- Web tools: we mainly offer three web-based tools, the EDC system, the eTMF, and the DICOM imaging platform, to provide more accurate data collection for soft tissue sarcoma trials.
In addition to the wide variety of services provided by Sofpromed, our CRO is also characterized by its responsiveness and client proximity. Small biotech companies usually prefer a business partner that clearly understands their specific needs and challenges, and capable of offering them personalized services. If this is your case, Sofpromed is the CRO partner you are looking for.
Sofpromed: An Excellent Partner for Conducting Clinical Trials for Soft Tissue Sarcoma
Sofpromed is a full-service clinical research organization (CRO) specialized in managing phase I-IV clinical trials in oncology. We are known for our technical proficiency and deep knowledge of a wide variety of cancerous diseases, including soft tissue sarcoma. Sofpromed helps to assure standards of quality for clinical research and focuses on delivering optimal outcomes for biotech companies.
Our target is to give the best support to biotech companies by providing them with competent staff of scientific vocation and training as well as the most suitable tools to meet all the requirements that they should fulfill to run a clinical trial for soft tissue sarcoma. Ultimately, by virtue of our client proximity, Sofpromed is a real CRO partner for small and mid-sized biotech companies planning to run oncology clinical trials worldwide in general, and in soft tissue sarcomas in particular.
If you need a CRO to conduct a soft tissue sarcoma clinical trial in the US, please contact us at info@sofpromed.com
References:
[1] Lazarides, Alexander L., Julia D. Visgauss, Daniel P. Nussbaum, Cindy L. Green, Dan G. Blazer III, Brain E. Brigman and William C. Eward. 2018. “Race Is an Independent Predictor of Survival in Patients with Soft Tissue Sarcoma of the Extremities.” BMC Cancer 18, 488. [Accessed 3 May 2022]
[2] American Cancer Society. “Key Statistics for Soft Tissue Sarcomas.” [Accessed 2 May]
[3] American Cancer Society. “Radiation Therapy for Rhabdomyosarcoma.” [Accessed 5 May]
[4] Nakano, Kenji and Shunji Takahashi. 2018. “Current Molecular Targeted Therapies for Bone and Soft Tissue Sarcomas.” International Journal of Molecular Sciences 19(3). [Accessed 6 May]