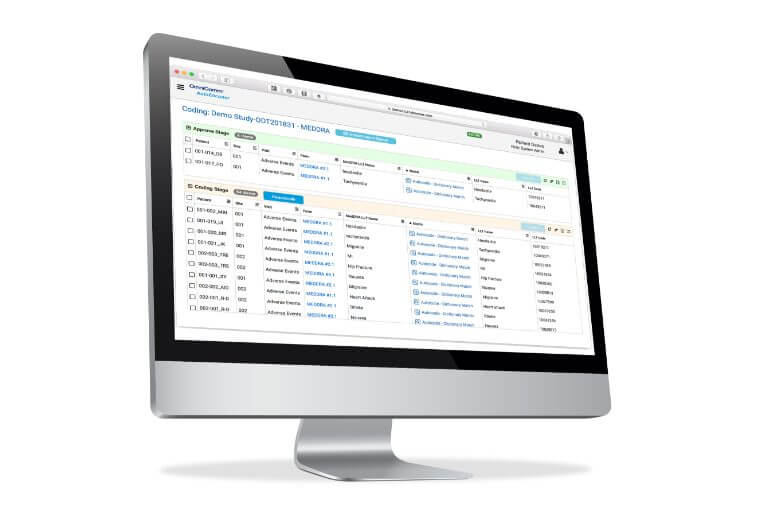
OmniComm’s TrialMaster® EDC: Streamlined CDISC SDTM Clinical Trial Data Submissions to the FDA
Deprecated: preg_replace_callback(): Passing null to parameter #3 ($subject) of type array|string is deprecated in /var/www/vhosts/sofpromed.com/httpdocs/wp-content/themes/Divi/includes/builder/feature/dynamic-content.php on line 1958