Why is data management so important in clinical trials? Sponsors should ensure the highest data quality in their research.
How are clinical trials conducted? What is a CRO? Read our articles and learn more about clinical trials and contract research organizations.
What is a Biometrics CRO?
Please contact us at info@sofpromed.com if you need a biometrics CRO for your clinical trial Clinical research and drug development have evolved significantly, with an increasing number of data...
Clinical Data Managers for Clinical Trials
If you need Clinical Data Managers for your clinical trials, please contact us at info@sofpromed.com How crucial and essential data management is in a clinical trial must be one of the most common...
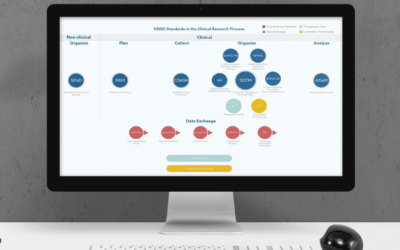
Why is CDISC CDASH Important?
If you need to implement CDISC CDASH in your clinical trial eCRF, please contact us at info@sofpromed.com Conducting a clinical trial involves managing a great amount of data, from the very...
Clinical Data Management Services in Clinical Trials
If you need clinical data management services for clinical trials, please contact us at info@sofpromed.com Clinical data management is a fundamental element for the success of a clinical trial....
CDISC Services for Clinical Trials
If you need CDISC services, contact us at info@sofpromed.com CDISC services are related to CDISC standards, one of the most important data management standards in the clinical research industry....
Does the FDA Require CDISC?
If you need CDISC services for clinical trials, please contact us at info@sofpromed.com Clinical trial data standardization is important when it comes to data management and clinical data...
Clinical Data Management, Biostatistics, and Statistical Programming Services for Clinical Trials
Sofpromed is a clinical research organization (CRO) providing cutting-edge clinical data management, biostatistics, and statistical programming services in SAS for phase I-IV clinical trials across...
Clinical Data Management Service Providers for Clinical Trials in the United States
Are you looking for clinical data management service providers for clinical trials conducted in the United States? IQVIA, Quanticate, Syneos Health, Linical, eClinical Solutions, ICON, and Sofpromed...
What is an Electronic Data Capture (EDC) System in Clinical Trials?
Contact us at info@sofpromed.com if you need an EDC system for your clinical trial. An electronic data capture (EDC) system —also referred to as electronic case report form (eCRF)— is a very...
How Much Does an Electronic Case Report Form (eCRF) Cost?
Click here to download a fully detailed Electronic Case Report Form (eCRF) quote An Electronic Case Report Form (eCRF) software —also known as Electronic Data Capture (EDC) system— is an essential...
CDISC SDTM Clinical Trial Data Submissions to the FDA: Frequently Asked Questions
What is SDTM? SDTM stands for “Study Data Tabulation Model”, and is a CDISC standard to structure (tabulate) clinical trial data to be submitted to regulatory authorities such as the Food and Drug...
Which Electronic Data Capture System Should I Use?
There are several types of electronic data capture (EDC) solutions that can be used to manage data in clinical trials. The selection of the data management tool will depend on the size of the study...
What is an eCRF?
An eCRF (electronic case report form) is a software system used to collect data in a clinical study. Commonly, eCRFs are web-based applications containing various data forms and fields designed to...
Reducing Data Queries Through Optimized eCRF Design
Cleaning data in clinical trials can be a tedious task when the electronic case report form (eCRF) has not been properly designed. Identifying and resolving discrepancies in data is a...